From the understanding of the indispensable importance of NAD+, medical scholars have discovered that its predecessor NAD+ precursor / booster (NMN or NR) can effectively increase the level of NAD+ in the body; up to now, in the known knowledge category, NAD+ cannot be stored stably at room temperature state, and cannot directly increase the NAD+ level in the body after oral administration.
We need to take NMN or NR to effectively increase NAD+ instead of taking NAD+ directly. However, there are many products that can be taken orally directly under the name of NAD+. Instead of listening to the marketing tactics in the market, let's take a closer look at what the US FDA has to say.
In 2017, the Pharmacy Compounding Advisory Committee of the US FDA made the following conclusions on whether Nicotinamide adenine dinucleotide (NAD+) should be included in effective oral drug ingredients:
Veto (Against)
The reason why the FDA vetoed NAD+ into effective oral drug ingredients is that NAD+ will decompose when it is above minus 20 degrees Celsius (-20oC); NAD+ will also decompose when it encounters alkali, sunlight, and moisture, and finally decomposes into nicotinamide riboside, Substances such as nicotinic acid and nicotinamide are collectively referred to as niacin. In other words, it is impossible for NAD+ to exist stably in a health supplement capsule stored at room temperature. From the moment it leaves minus 20 degrees Celsius, it is no longer NAD+, but a substance such as niacin.
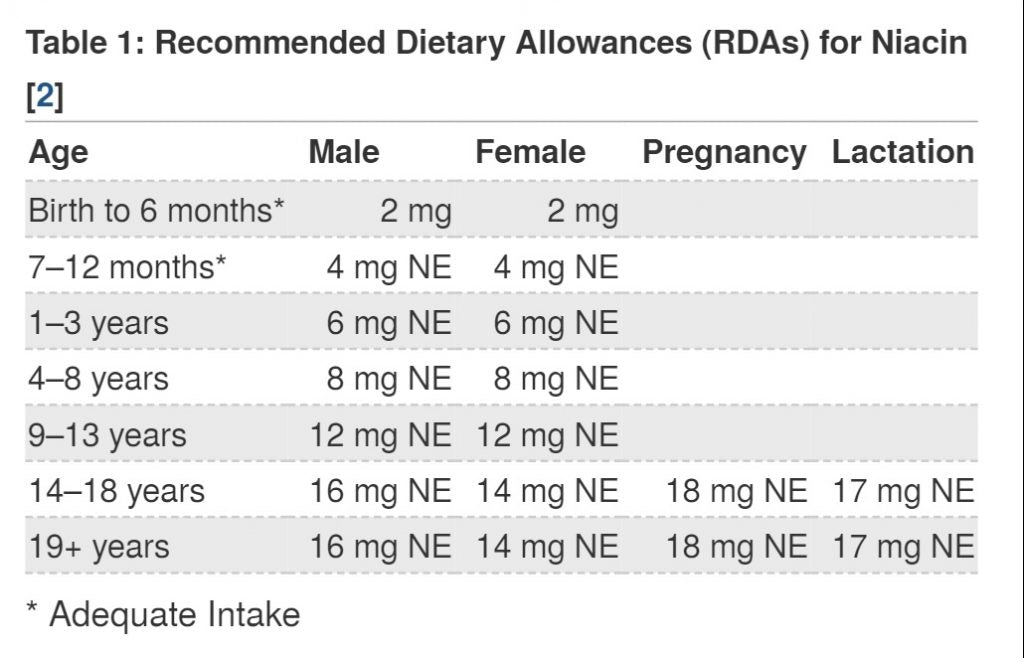
The related potential worry is that the human body has an upper limit for the absorption and intake of niacin, and the recommended dietary allowance for adults should not exceed 15mg niacin. (As shown below)

NAD+ decomposed nicotinmade, if the human body absorbs more than 1000mg per day, it will cause gastrointestinal adverse events (gastrointestinal adverse events); if the intake dose is greater than 3000mg, it is more likely to cause liver toxicity.
According to the FDA, the intake of 5mg NAD+ is similar to the recommended dietary allowance of 15mg of niacin for adults.
It has been clinically shown that taking high doses of Niacin may also cause skin redness, fever, burning pain, itching and other symptoms, which is called niacin flush. Professor David Sinclair of Harvard University has publicly mentioned that we do not want to take high doses of nicotinamide because it will inhibit the longevity genes Sirtuins and PARP, hindering the repair of gene damage. Prof. David Sinclair: "Nicotinamide which is something you don't want to take high doses of because we've shown in my lab many years ago that nicotinamide will inhibit the sirtuins, and PARP as well, and interfere with DNA repair."
Just imagine if it is feasible to take NAD+ directly, why do many scholars, Professor Shinichiro Imai, and Prof. David Sinclair mention NMN instead of NAD+ in their research and writings? Please be careful not to fall into the trap of businessmen and delay the health of yourself and your family.
At the same time, the United Kingdom, Europe, Japan and other places have not listed NAD+ in the effective pharmacopoeia; based on the above considerations, the FDA has rejected the inclusion of NAD+ as an active drug ingredient under Regulation 503A of the US Federal Food, Drug, and Cosmetic Act.
The author recommends that if you are planning to take it, you must understand its ingredients and content clearly when you are already taking food that advertises NAD+ as a direct supplement. If the body reacts at all, stop taking it immediately and consult your doctor immediately.
Reference:
Bulk Drug Substances Used in Compounding Under Section 503A of the FD&C Act
COMPOUNDERS UNDER SECTION 503A OF THE FD&C ACT: QUALITY STANDARDS AND FDA FINDINGS, Pharmacy Compounding Advisory Committee Meeting May 8, 2017
Niacin - Fact Sheet for Health Professionals, National Institutes of Health, US Department of Health & Human Services
At this time, you need Chuanyu NMN